KETOSIS: Amino acid nutrition, different types of ketosis and related inflammatory conditions

Dear Farmer,
In this third issue of the Tecnozoo update, we will address a highly relevant topic: ketosis, a metabolic condition affecting a significant number of cows in the immediate post-partum period. It is characterised by an increase in ketone bodies (acetoacetate, acetone and β-hydroxybutyrate, the latter being the most prevalent, accounting for approximately 70% of total ketone bodies) in body fluids (blood, urine and milk). Ketosis is often associated with various health issues (fever, metritis, abomasal displacement, mastitis, etc.) and zootechnical problems (marked reduction in appetite, significant weight loss, low milk production, and a generally depressed and lethargic condition), resulting in serious economic losses.
But where does ketosis originate, and what is its relationship with pre-partum inflammatory states?
Let us analyse the most recent studies on this topic and energy management during the transition period, with insights provided by our technician Pierantonio Boldrin.
Where does ketosis come from? Management of negative energy balance during the transition phase
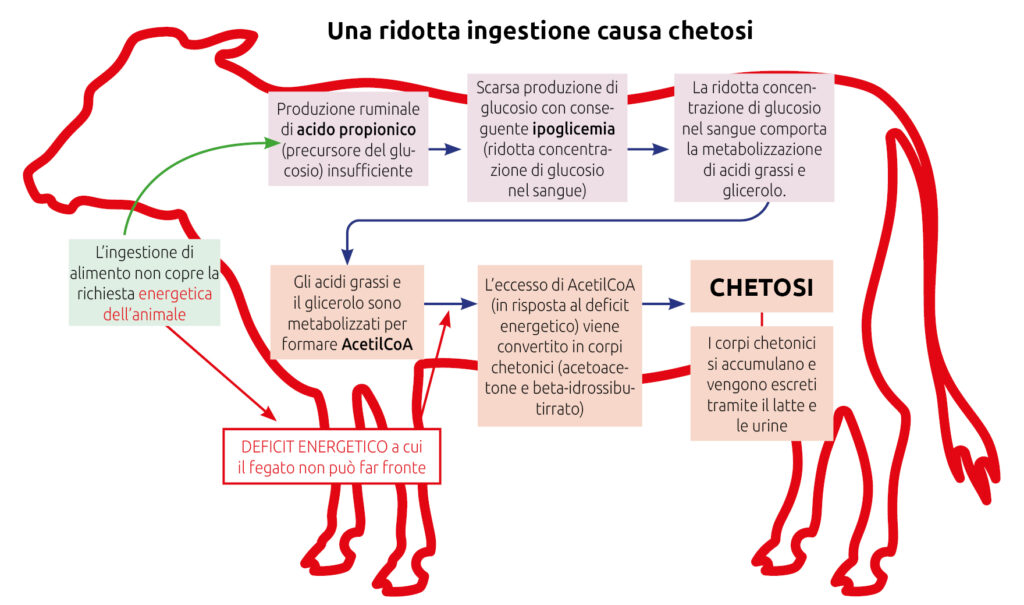
Ketosis in dairy cows is defined as a metabolic condition in which the concentration of ketone bodies in the blood exceeds the physiological threshold of 1.2 mmol/L for β-hydroxybutyrate. To understand this increase, it is essential to identify its origin. Ketone bodies may have an exogenous origin (from the diet) or, more commonly, an endogenous origin (from hepatic metabolism).
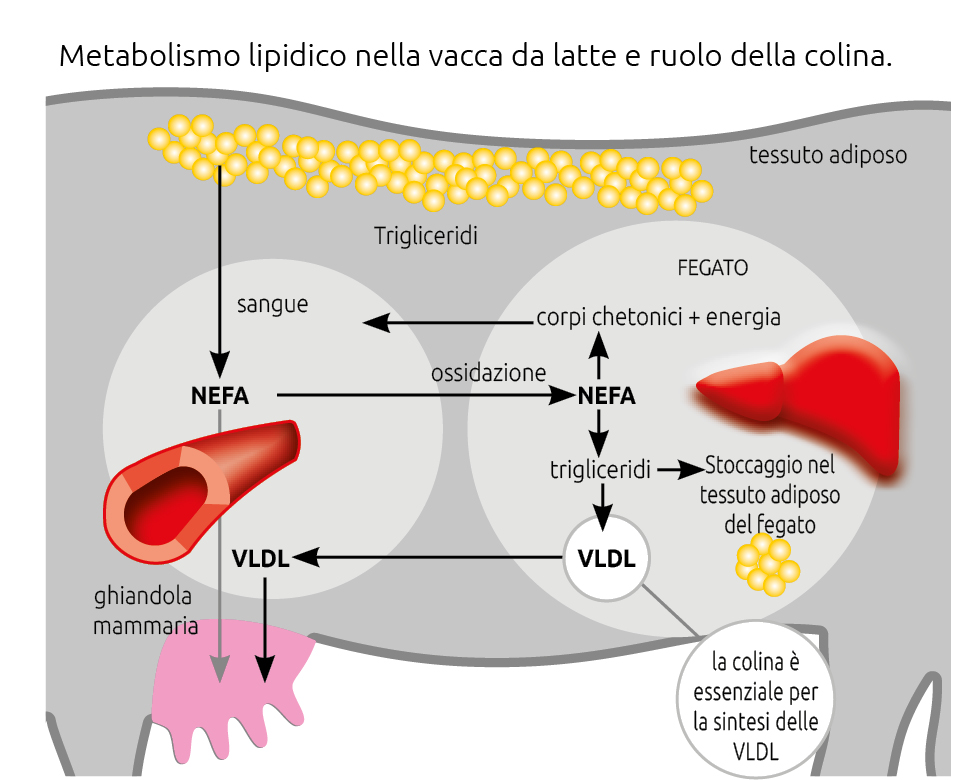
Ketosis is generally considered to arise from a state of negative energy balance (NEB) occurring at the onset of lactation, due to an energy deficit between the demands of milk production and the cow’s intake capacity in the post-partum period. This condition triggers lipomobilisation from body reserves, with triglycerides released as non-esterified fatty acids (NEFA), which enter the bloodstream and reach the liver. Under normal conditions, these fatty acids are re-esterified and released back into circulation as VLDL.
When hepatic re-esterification capacity is insufficient (due to excessive lipomobilisation and/or a deficiency of lipotropic factors such as choline), triglycerides accumulate in the liver, leading to hepatic lipidosis and impaired liver function, with a further reduction in glucose availability due to decreased gluconeogenesis.
In addition, if mitochondrial oxidative capacity is compromised by a reduced availability of intermediates of the tricarboxylic acid cycle—caused by energy deficiency—ketone body production increases. If these ketone bodies are not utilised by peripheral tissues, they accumulate in the bloodstream.
Ketone bodies do represent an alternative energy source to glucose (for example, the brain can use them as they cross the blood–brain barrier, unlike NEFA). However, their accumulation has negative effects on appetite regulation, immune function, and oocyte and embryo development. This explains the poorer health and production outcomes observed in ketotic cows compared to non-ketotic animals.
Nevertheless, the NEB-based aetiology of ketosis is not entirely convincing, since nearly all cows experience an energy deficit at the start of lactation, yet only some develop ketosis. This raises the question: are elevated ketone bodies the cause of ketosis-related diseases and reduced performance, or are they a metabolic response to underlying causal factors (such as stress), which then lead to these consequences?
Classification of ketosis
Clinical, subclinical and classification by time of onset
There are essentially two different approaches to classifying ketosis, based either on the quantity of ketone bodies present in the blood or on the origin and time of onset of the ketotic condition. Based on blood ketone body levels, ketosis is classified as subclinical ketosis (SK) at blood β-hydroxybutyrate values of ≥ >1.2 mmol/L and clinical ketosis (CK) at values above 2.9 mmol/L.
The clinical form of ketosis (CK), unlike SK, is accompanied by loss of appetite and body weight, low milk production and dry faeces.
Classification by time of onset and origin identifies three types of ketosis:
Type I (primary ketosis): occurs in the first weeks of lactation, analogous to type I diabetes in humans. It is characterised by hypoinsulinaemia caused by chronic glucose deficiency, as glucose is preferentially used by the mammary gland.
Type II (secondary ketosis): occurs immediately around calving, similar to type II diabetes in humans. It is characterised by hyperinsulinaemia and hyperglycaemia and is typical of overconditioned cows at calving, with excessive lipomobilisation, hepatic lipidosis and impaired hepatic gluconeogenic and immune function.
Type III ketosis: results from ingestion of ketone bodies through diets containing altered silages derived from undesirable clostridial fermentations with high butyric acid levels.
Ketosis: not only negative energy balance
The hypothesis of chronic inflammation as a cause of ketosis and related disorders
To assess the causal link between chronic inflammation and ketosis, we must consider whether inflammation precedes ketosis, what triggers the inflammatory event, and how it contributes to ketosis development.
An interesting study (Zhang et al., 2016) showed that cows developing clinical ketosis post-partum already had significantly elevated levels of major pro-inflammatory markers (IL-6, TNF, SAA and lactate) at −8 and −4 weeks pre-partum compared to non-ketotic cows.
This suggests that inflammation precedes and predisposes to ketosis. Potential pre-partum inflammatory triggers include mammary infections, overcrowding, heat stress and digestive disorders, particularly ruminal and/or intestinal acidosis. These conditions are often associated with endotoxin (LPS) release, exacerbating inflammation and altering glucose and lipid metabolism, thereby triggering a cascade leading to increased ketone body production.
Other studies have shown reduced chemotactic capacity and oxidative efficiency of neutrophil granulocytes during ketosis, likely due to reduced glucose availability and calcium deficiency. Hypocalcaemia frequently accompanies hepatic lipidosis and ketosis. Elevated inflammatory biomarkers such as LPS further confirm the strong inflammatory component of ketosis.
These mechanisms explain why ketotic cows are 3 to 8 times more likely to develop diseases such as retained placenta, metritis, mastitis and abomasal displacement, and have a significantly higher culling risk in the first 60 days post-partum compared to healthy cows.
How can we intervene?
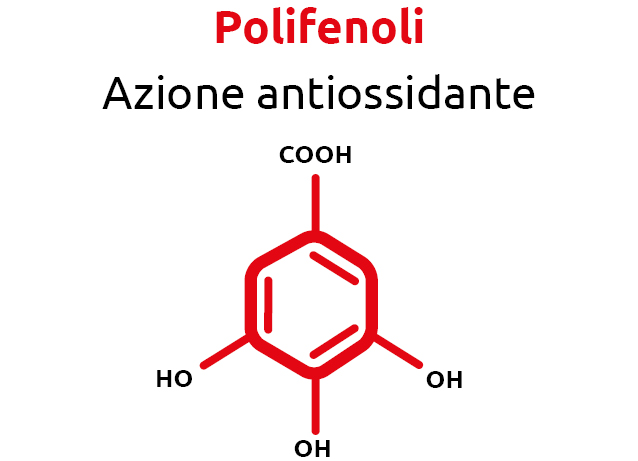
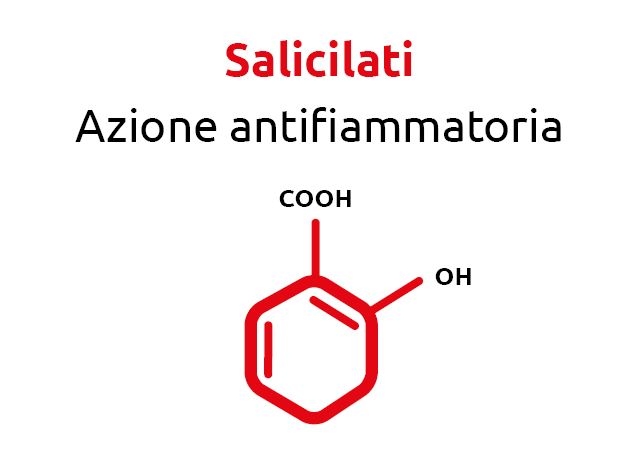
Once again, the answer comes from nature. Natural compounds such as polyphenols and salicylates, when properly selected and dosed, can rebalance oxidative and inflammatory conditions. These principles were used in the formulation of Normoterm Orac.
Numerous studies have demonstrated the effectiveness of natural phytocompounds—such as polyphenols and salicylates—on animal metabolism, acting as antioxidants and attenuating inflammatory states.
One of the richest sources of polyphenols is lignin derived from Pinus taeda. Lignin is a complex polymer composed of phenylpropanoid units (coniferyl, coumaryl and sinapyl alcohols). Their combination forms lignans, which in plants serve defensive and antioxidant functions. In mammals, lignans are modified by intestinal bacteria, absorbed and subjected to enterohepatic circulation.
Mechanism of action
A polyphenolic concentrate derived from extraction and partial hydrolysis of cellulose from Pinus taeda wood exerts the following actions when included in the diet:
1. Free radical scavenging
2. Antibacterial activity
3. Antiviral activity
4. Antimycotoxin activity
5. Antidiarrhoeal activity
Salicylic acid, which is the active compound of aspirin in its acetylated form, is a substance that was first extracted from the bark of the weeping willow.
This compound, produced by plants to protect themselves from chemical, physical and biological attacks, acts as an anti-inflammatory and is useful in various febrile conditions as an antipyretic or mild analgesic.
Mechanism of action
1. Reduction of prostaglandins via cyclooxygenase inhibition
2. Antioxidant effect via haptoglobin
3. Reduction of body temperature through action on thermoregulatory neurons
NORMOTERM ORAC
Complementary mineral feed for cattle and buffaloes.
- Supplementation with antioxidant plant-derived compounds from the Normoterm line helps counteract alterations caused by environmental conditions, maintaining physiological balance and supporting performance.
- Contains White Willow, Meadowsweet, Red Vine and Hop.
Nutritional management of energy metabolism during transition: Return on investment in the control of ketosis and hepatic lipidosis at calving and their consequences.
In the first weeks post-partum, nutrient requirements for milk production increase rapidly, along with the risk of ketosis. Preventive management focuses on body condition score before calving and improving dry matter intake post-partum.
It is possible to implement a series of management and nutritional measures to reduce the onset and impact of subclinical and clinical ketosis in cattle. Preventive management focuses on body condition score (BCS) before calving and on improving dry matter intake (DMI) after calving.
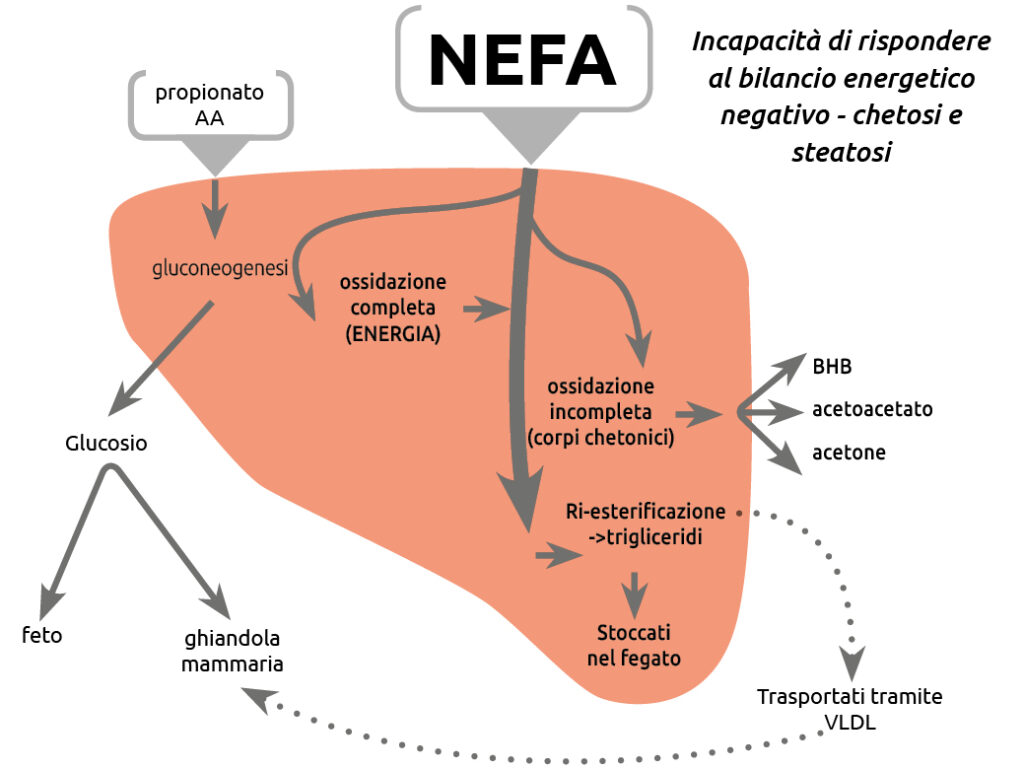
By briefly describing the metabolic fate of fatty acids mobilised from adipose tissue during negative energy balance (NEB), the usefulness of implementing specific additives during the transition phase becomes clear.
Non-esterified free fatty acids (NEFA) reach the liver via the bloodstream and, within the hepatocyte, first bind to coenzyme A and subsequently, as acyl-CoA, to carnitine. Carnitine transports them into the mitochondria, the intracellular organelles of the hepatocyte where fatty acid oxidation and energy production occur.
Complete oxidation, however, depends on the availability of energy precursors of the oxidative cycle, in particular oxaloacetic acid, derived from carbohydrate metabolism. In the absence of sufficient oxaloacetate, partial rather than complete oxidation occurs, and acetyl-CoA molecules—originating from fatty acid oxidation or from pyruvic acid produced by amino acid or carbohydrate catabolism—combine to form ketone bodies. The condensation of two acetyl-CoA molecules produces acetoacetate (AcAc), from which acetone and β-hydroxybutyrate (BHBA) are subsequently formed.
An excessive supply of fatty acids at the hepatic mitochondrial level, in the absence of adequate levels of oxaloacetic acid, therefore leads to a high production of ketone bodies which, being unable to be utilised by body tissues, accumulate in the blood, giving rise to ketosis and a vicious circle of further worsening of NEB which, if not interrupted, progresses to the loss of the animal; and if a deficiency of carnitine occurs at the hepatic level, the NEFA reaching the liver are unable to enter the mitochondria to be oxidised, resulting in their accumulation within hepatocytes and giving rise to steatosis or hepatic lipidosis, a pathological condition that is often associated with type II ketosis in the immediate post-partum period.
FOCUS: amino acid nutrition during the transition phase
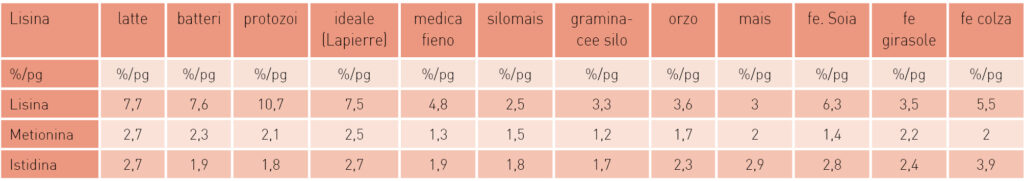
In recent years, research on dairy cow ration formulation has become increasingly refined, and one of the latest frontiers is represented by amino acid nutrition. In monogastric livestock, this science has been applied for many years, but in polygastric animals the forestomachs make responses less predictable. It is undeniable that cows also have so-called essential amino acids, meaning those they cannot synthesise themselves and therefore must be supplied through the diet. Fortunately, ruminal fermentations can partially compensate for this, but especially in high-producing cows the requirement cannot be fully met. For this reason, certain limiting amino acids are identified, among which the most important are methionine, lysine and histidine.
This type of rationing must meet two essential requirements: amino acids must maintain a correct balance among themselves and, if supplemented, must be rumen-protected to prevent degradation in the rumen.
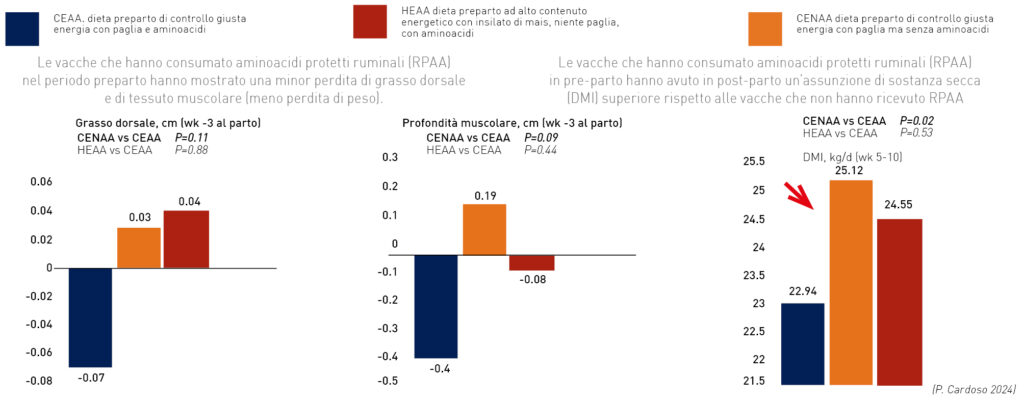
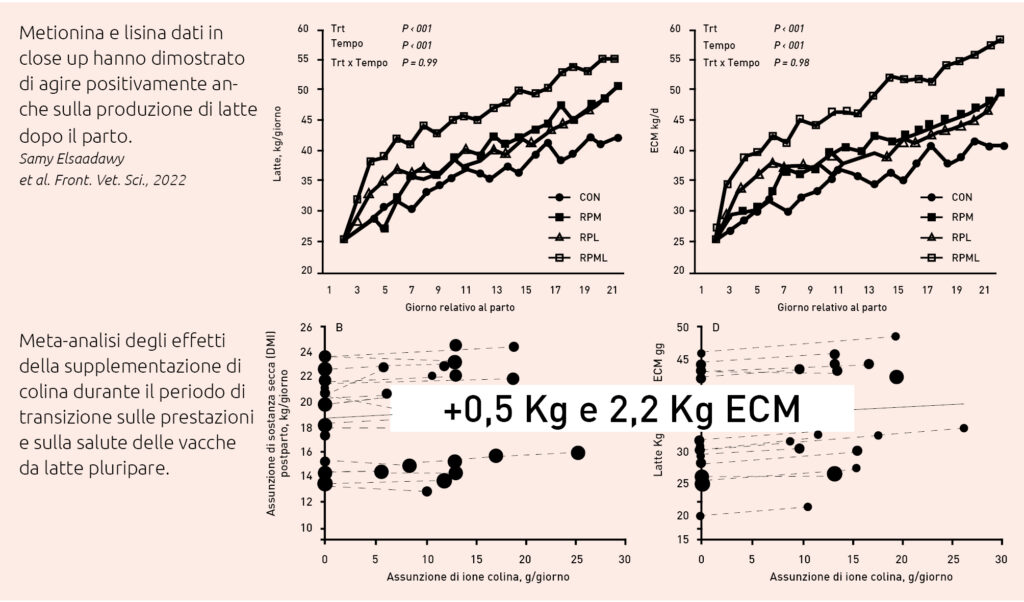
One phase of the production cycle in which a particular benefit from the use of amino acids has been observed is the late dry period and the post-partum phase.
PERIPARTUM CARE
Complementary feed for dairy cows
- Contributes to increasing metabolizable protein.
- Rumen-protected lysine and methionine to meet requirements and modulate the correct ratio between them.
- RP choline for its well-known anti-ketotic effect and beyond.
- Supplies omega fatty acids from flaxseed.
- Magnesium and sodium, often deficient in the ration.
- Vitamins and essential oils targeted to protect the intestinal mucosa and limit the disruption of tight junctions.
Ketosis and milk production
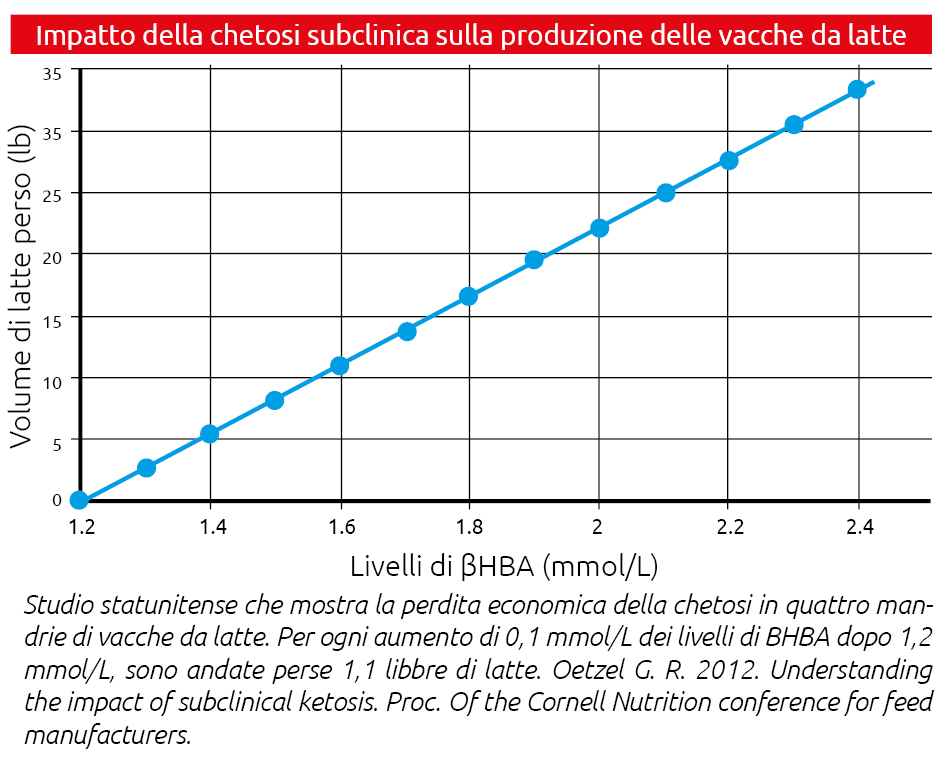
Milk production is correlated with the amount of lactose available, which is directly proportional to hepatic function and the glucose levels dependent on it. Reduced glucose production at the beginning of lactation during ketosis lowers prolactin levels and the optimal maturation of mammary gland secretory cells, resulting in a reduction in milk production capacity according to the severity of ketosis. Conservative estimates suggest that losses may range from 1 to 3 kg at peak lactation and may exceed 350 kg of lost production over the entire lactation (Ospina et al., 2010).
Ketosis and reproduction
The negative effect of NEB occurring in early lactation on subsequent reproductive performance is well documented, and it is now well established that the physiological basis of this condition is the disruption of the hypothalamic–pituitary–ovarian axis (Butler, 2003). Both the duration and the severity of NEB have been associated with increased growth hormone concentrations and decreased insulin and IGF concentrations; this leads to a state of “metabolic hunger” due to glucose deficiency in hypothalamic neurons of the arcuate nucleus, with severe repercussions on GnRH synthesis and LH pulsatility, directly reducing follicular competence and its response to circulating gonadotropins (Lucy, 2001; Butler, 2003).
For this reason, in cases of NEB and reduced LH peak amplitude, anovulatory cows and delayed resumption of ovarian cyclic activity occur, along with an increased incidence of cystic ovarian disease and a lower probability of pregnancy at first insemination (Opsomer et al., 2000; Ospina et al., 2010; McArt et al., 2012). In one study (Walsh et al., 2017), pregnancy probability was reduced by 20% in cows diagnosed with subclinical ketosis in the first or second week after calving.
However, cows above the subclinical ketosis threshold in both the first and second weeks post-partum had a 50% lower probability of becoming pregnant after the first insemination. Similarly, the calving-to-conception interval increased to 124 days in cows showing elevated BHBA concentrations in the first week after calving, and even to 130 days in cows with subclinical ketosis in both the first and second weeks post-partum, compared with cows that never showed elevated BHBA concentrations and had a shorter calving-to-conception interval.
Economic impact of ketosis
What has been described regarding the consequences of ketosis on milk production, fertility, increased susceptibility to related diseases and increased culling risk highlights the considerable economic importance of ketosis as a cause of reduced profitability in dairy farming. Ketosis generates direct costs related to specific treatments, labour time devoted to ketotic animals (monitoring and therapies), and milk losses due to reduced production capacity; but also indirect costs, even greater, resulting from concomitant diseases favoured by ketosis such as retained placenta, metritis, abomasal displacement, increased culling rate and reduced conception rate combined with an increase in days open.
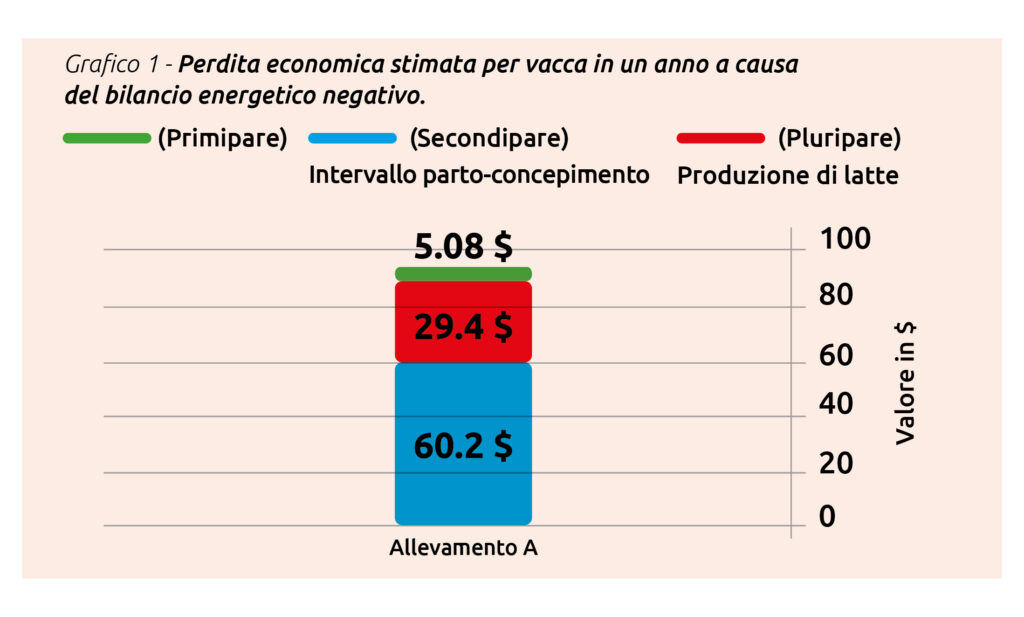
The cost of these events varies between primiparous and multiparous cows, with a conservative average value of approximately €85 in primiparous cows, rising to more than double (€180) in cows with multiple lactations.
Figure 1 shows the economic losses for primiparous (in green), second-parity (in blue) and multiparous cows (in red), expressed in dollars per cow per year, in a herd where early monitoring and treatment of ketosis are not implemented.
KETONIC
Dietetic complementary feed for reducing the risk of ketosis in dairy cows.
Start safely!
Why use Ketonic to reduce the risk of ketosis?
- Propylene glycol –> rapidly available energy
- Glycerol – > slower energy release with prolonged action
- Sugars –> improve rumen activity
- Vitamin B12 –> synergistic action with propylene glycol
- Niacinamide –> limits fat mobilisation
- Rumen-protected choline – >supports hepatic tissue in lipid metabolism
- Bypass methionine – >contributes to endogenous choline synthesis
- L-carnitine –> transports fatty acids into mitochondria to be used as an energy source
In conclusion, after analysing the metabolic dynamics, not only nutritional, that can lead to ketosis, it can be stated that investing in the health of our cows always makes sense from an ethical point of view, but in the case of this pathology it also becomes essential from an economic perspective, with ROIs that can reach values of up to 1:5.
COW DRENCH AG 3
Complementary feed for dairy cows
- Provides readily available calcium
- Supports rehydration and the supply of Na, K and Cl
- Sugars and proteins for immediate ruminal utilisation
- Highly palatable
To stay up to date with all our latest news, follow us on Instagram or on Facebook.